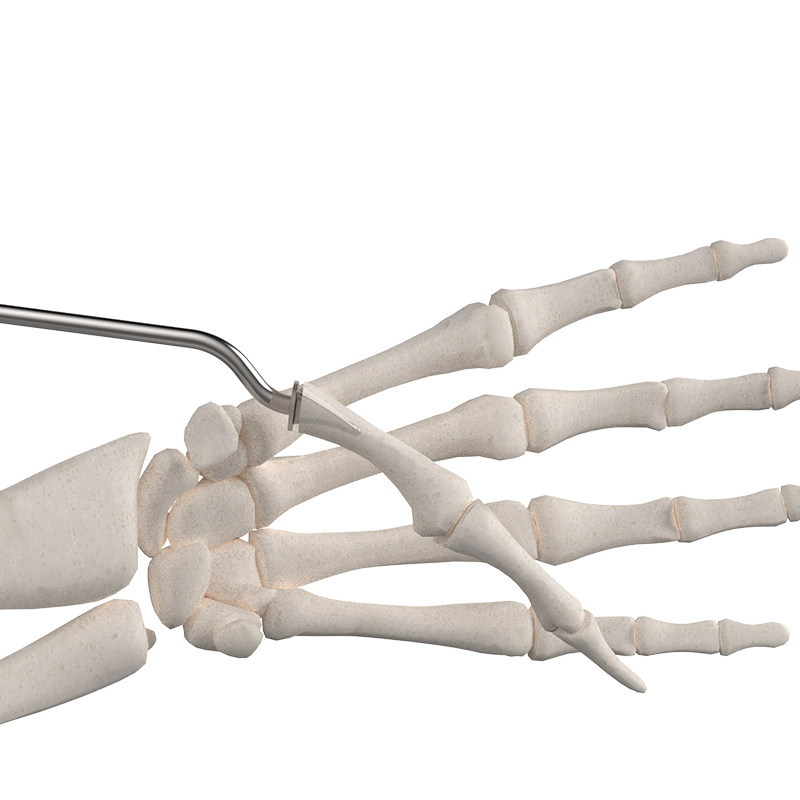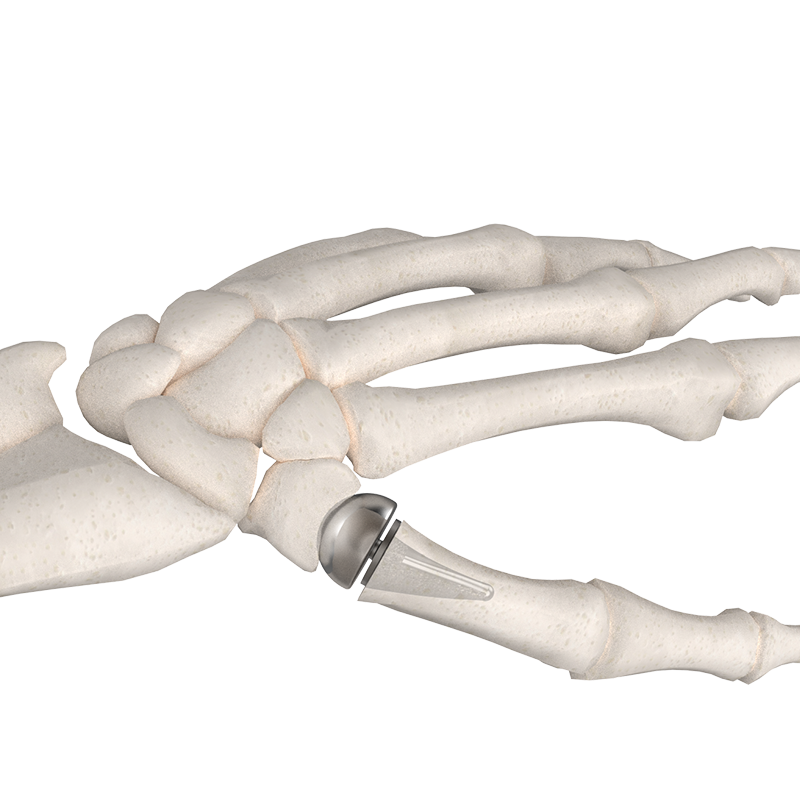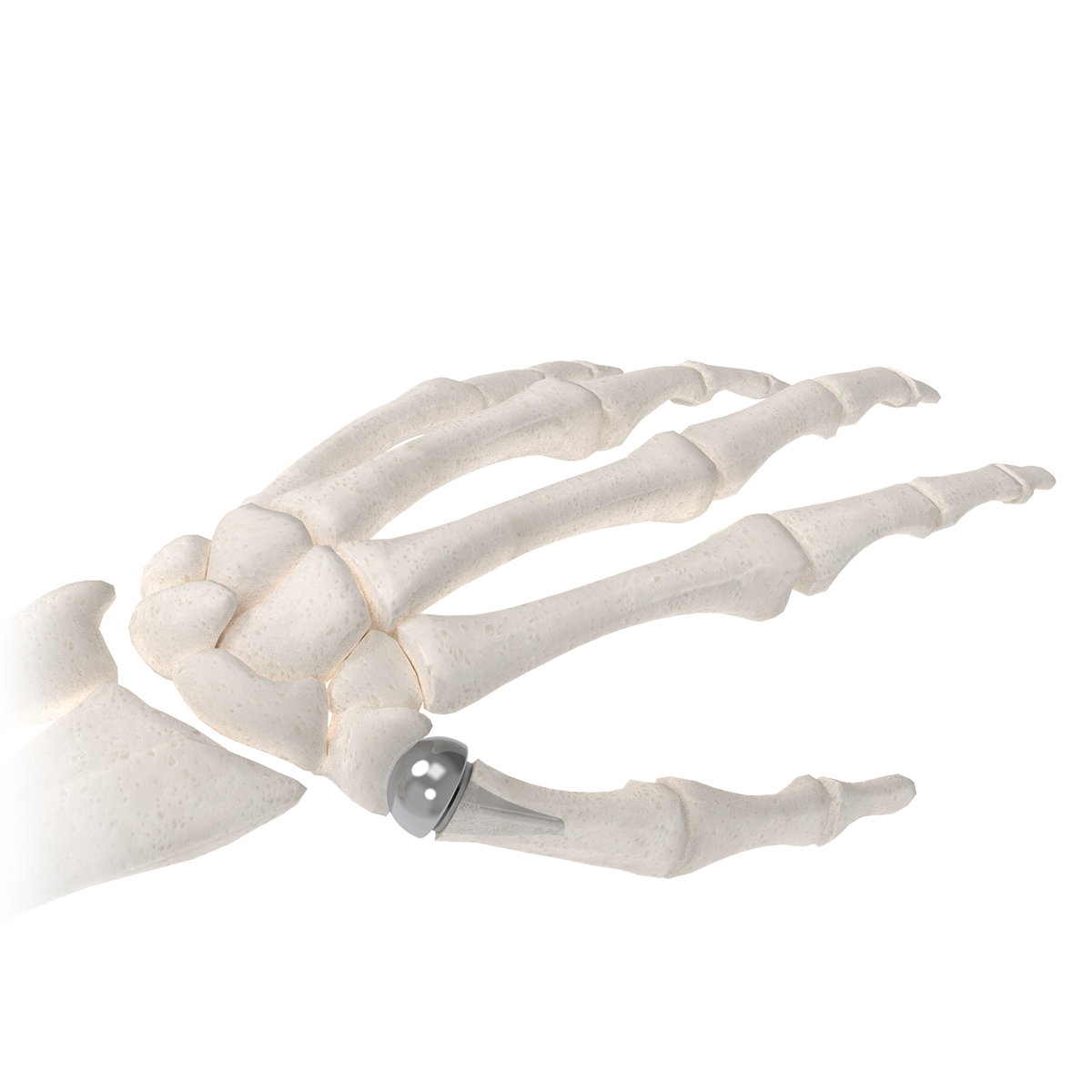
The BioPro Modular Thumb Implant, for your patients that demand more.
The BioPro Modular Thumb Implant is the only two-piece hemiarthroplasty system specifically designed for CMC arthritis. With more than 20 years of clinical use and published 94% survivorship at six years, the system has become a trusted alternative to traditional trapeziectomy-based procedures.
Documents
| Title | View | Type | hf:tax:doc_type |
|---|
Backed by 20 years of clinical use.
A review of 265 patients from three different published clinical studies.
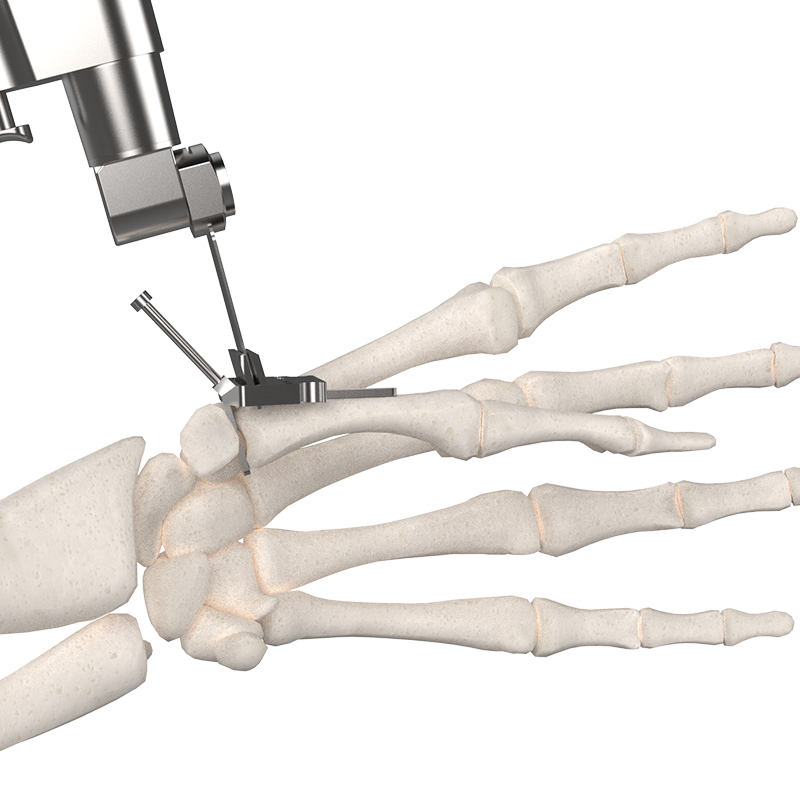
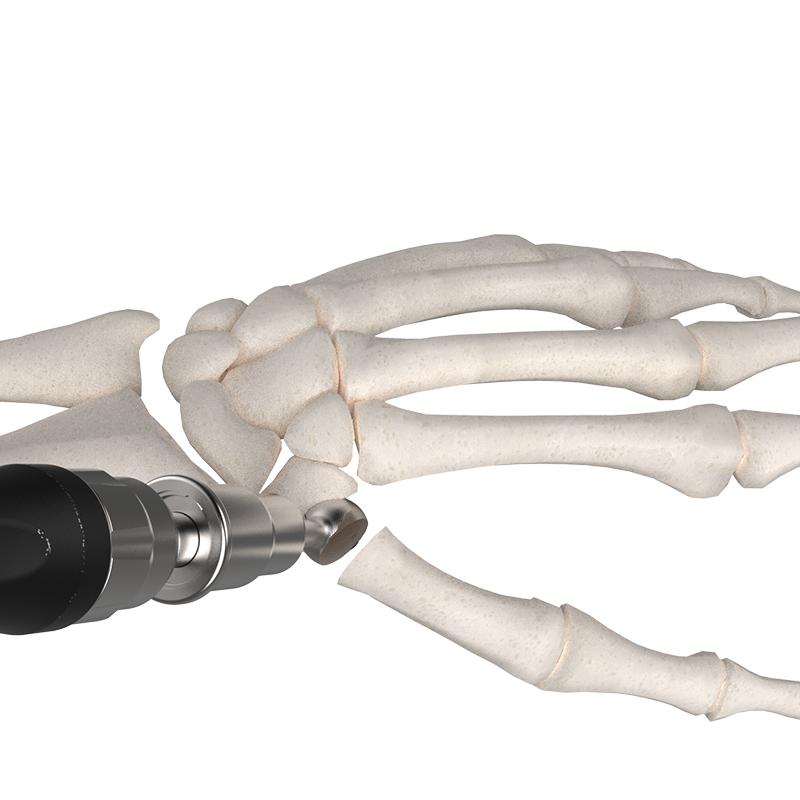
How it works

Postoperative Remodeling
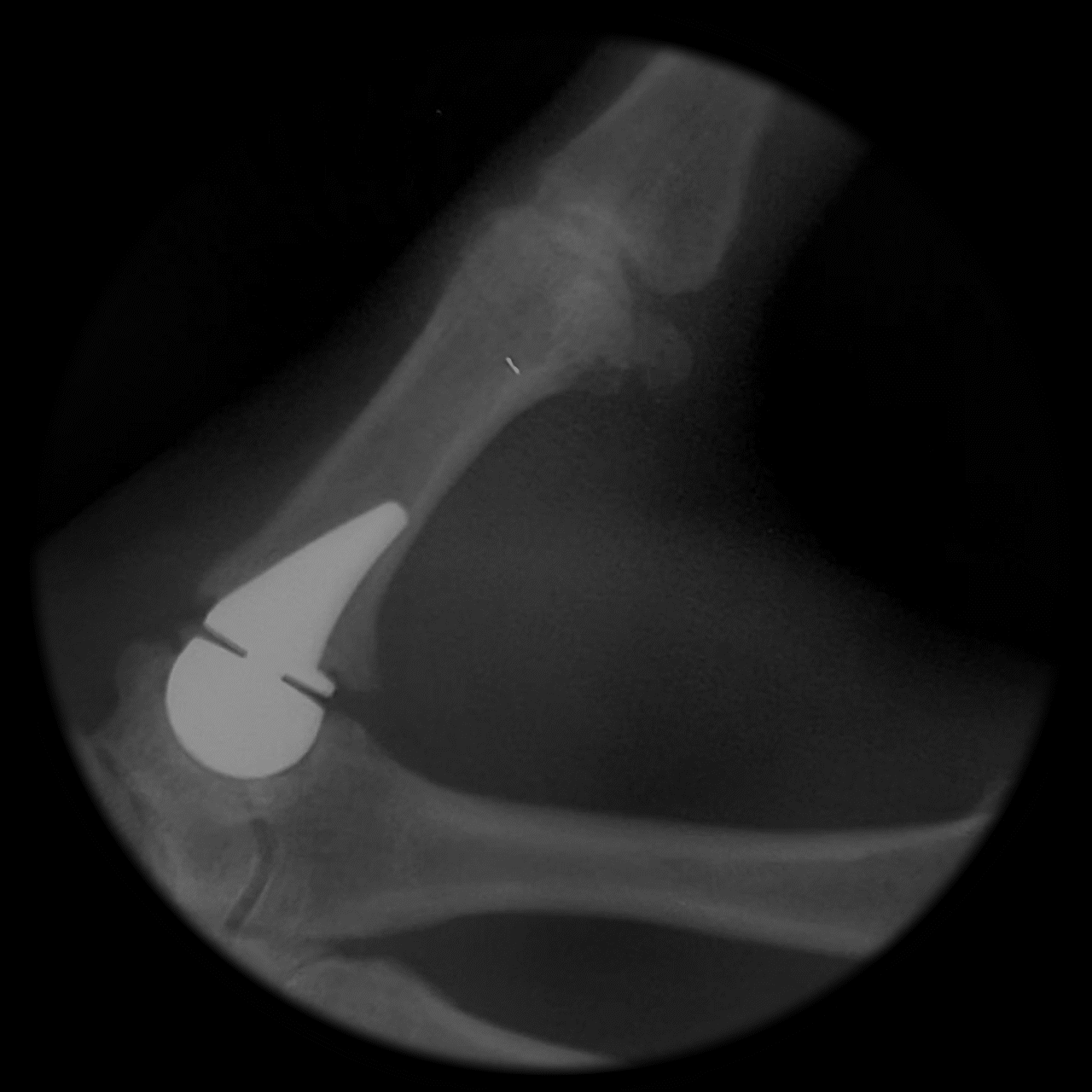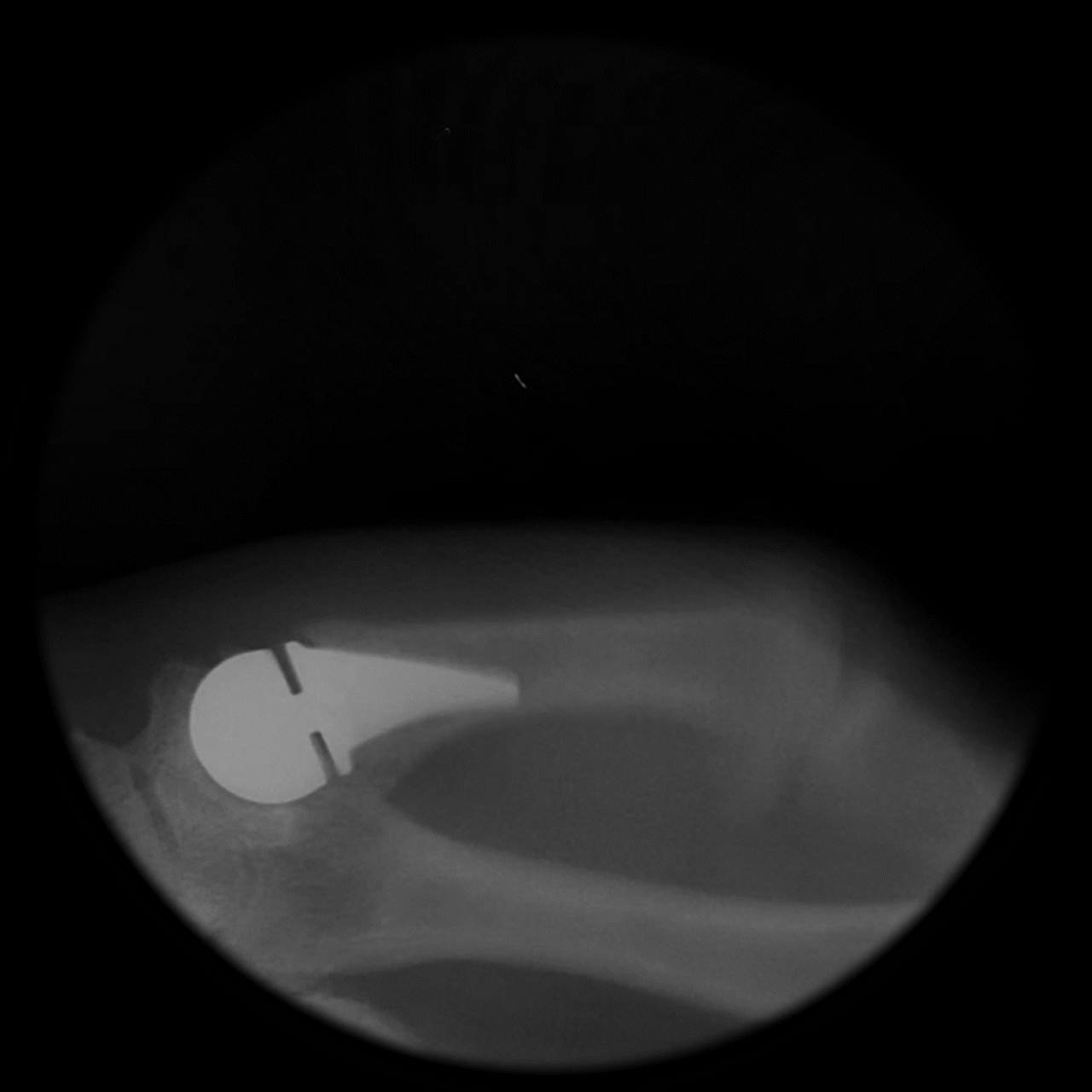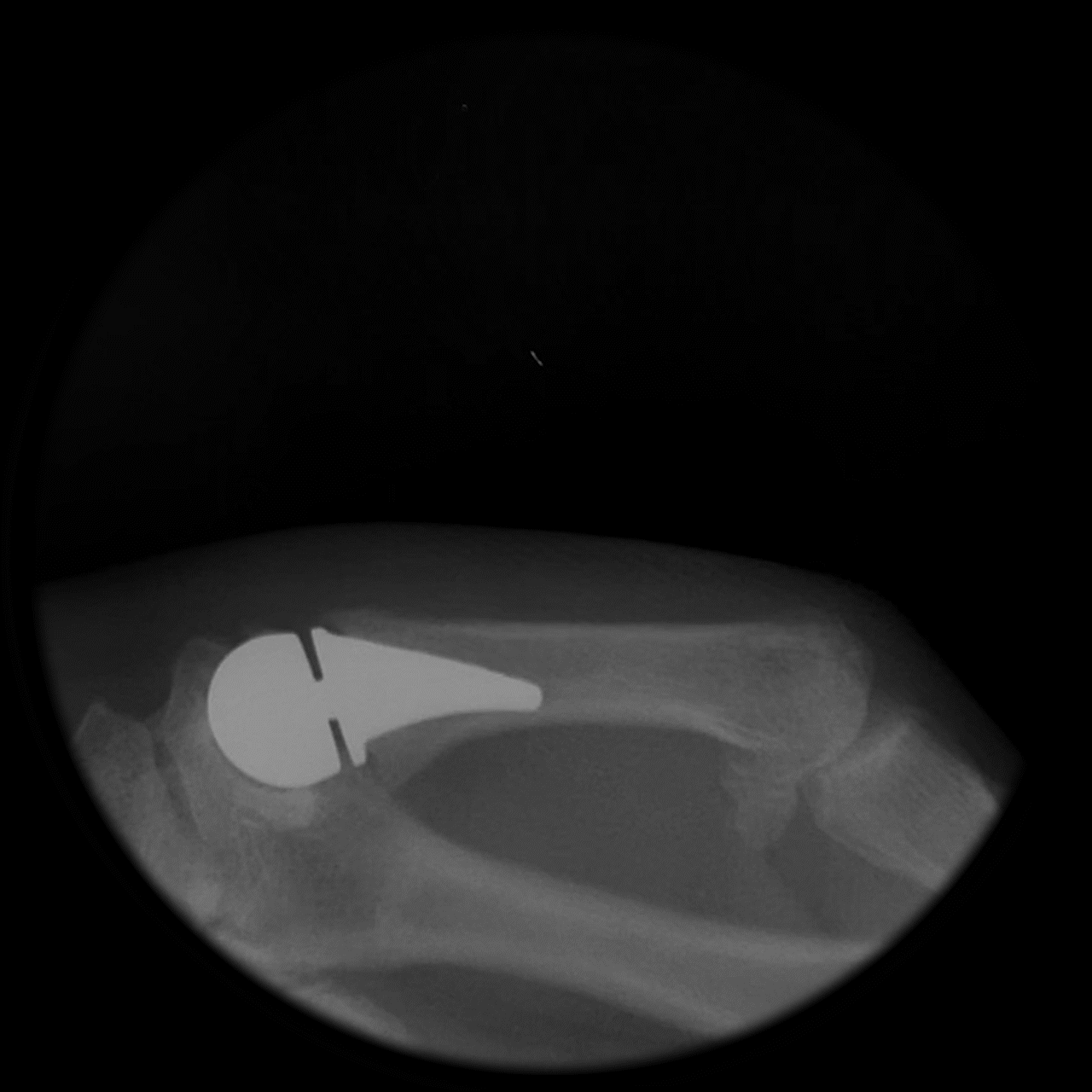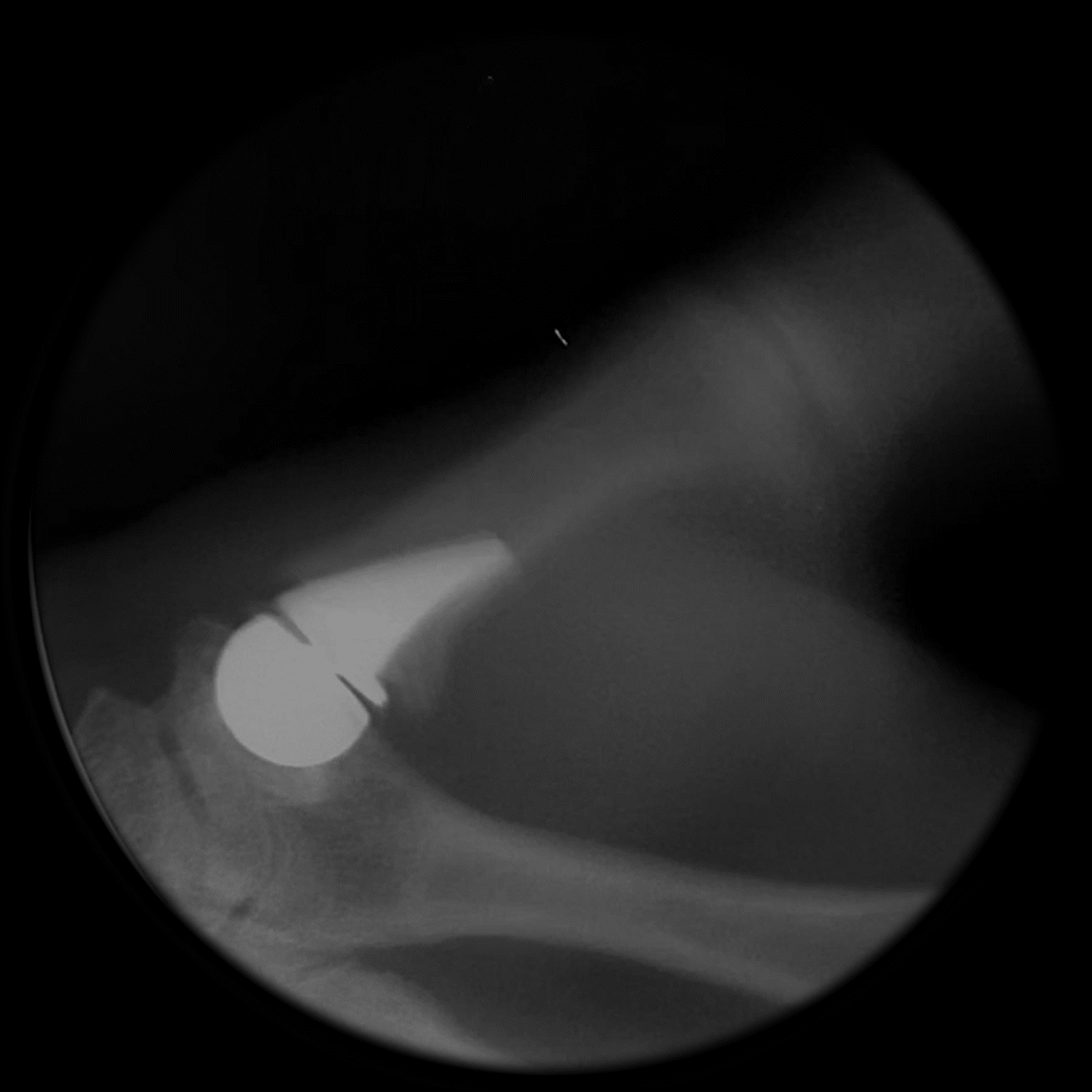
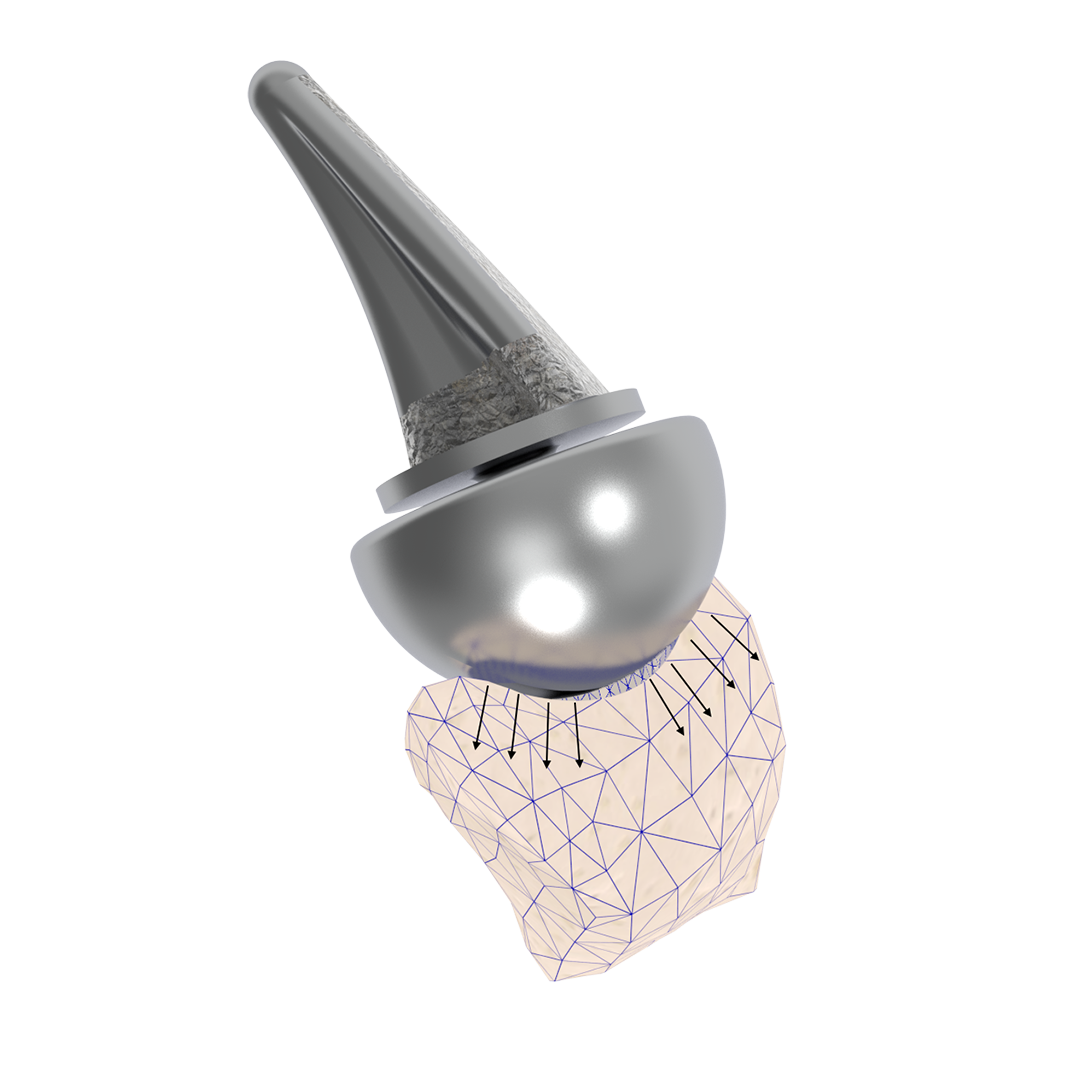
Following implantation, a predictable remodeling process occurs within the trapezium. During the first postoperative year, radiographs typically show gradual transformation of cancellous bone into a more sclerotic, dense structure as a result of physiologic loading.
This process leads to the formation of a congruent, stable socket that conforms to the implant head, creating a durable articulation surface and supporting long-term joint stability and function.
What patients are saying…
Patients are seeking alternatives to traditional procedures due to the long recovery times and limited revision options available. Our data proves that patients previously treated with an LRTI on their contralateral hand prefer the implant over the LRTI procedure.1 Listen to Ruth share her experience after receiving both an LRTI and a BioPro Modular Thumb Implant.
What surgeons are saying…
Leading surgeons are discovering the benefits of thumb joint replacement. The “burn no bridges” approach, allows surgeons to treat their patients earlier, with predictable results. Backed by 20 years of clinical experience, you can be confident you are treating your patients with a proven implant.

Tech Specifications
What’s Included
BioPro provides a reusable loaner instrument kit and (2) of each sterile packaged implants. Download the Bill Only Forms in the Document section for more ordering information.
Sizing
There are 48 total implant configurations. Head diameters of 12 mm, 13 mm, 14 mm, and 15 mm provide full trapezial coverage, each available with 0 mm, +2 mm, and +4 mm offsets to optimize joint tension. Four stem sizes—7.5 mm, 8.5 mm, 10 mm, and 11.5 mm—are offered, with +4 mm revision stems available for each size.
Material
The standard implant is manufactured from cobalt chrome, a highly biocompatible and durable material. A titanium version is available for use in patients susceptible to nickel chromium allergies.
Coating
The stem is coated with titanium plasma spray to improve biological fixation.
Videos
Find My Rep
REFERENCES
1.Pritchett JW, Habryl LS. A promising thumb Basal joint hemiarthroplasty for treatment of trapeziometacarpal osteoarthritis. Clin Orthop Relat Res. 2012 Oct;470(10):2756-63. doi: 10.1007/s11999-012-2367-7. Epub 2012 May 15. PMID: 22585348; PMCID: PMC3442003.
2.Chung J, Albino-Hakim S, Samuels K, Bodansky D, Badia A. The BioPro Thumb Carpometacarpal Hemiarthroplasty: Case Series and Surgical Technique. Tech Hand Up Extrem Surg. 2024 Mar 1;28(1):26-32. doi: 10.1097/BTH.0000000000000456. PMID: 37747076.
3.Alexander T Goodsett; Daniel C Jupiter; Navneet K Venugopal; John J Faillace.Intermediate-Term Follow-Up of Biopro® Modular Thumb Implant for Carpometacarpal Arthritis. (2016). Journalonsurgery.org. https://www.journalonsurgery.org/articles/js-v4-1147.html